Articles from AccurKardia

AccurKardia, an ECG analytics software company focused on establishing ECG as a broad biomarker, today announced a multi-year agreement with Specialized Medical. Under this partnership, AccurKardia’s FDA-cleared Class II software as a medical device (SaMD), AccurECG™ 2.0, is actively powering Specialized Medical’s state-of-the-art cardiac monitoring operations, which currently serve more than 20 clinics, medical practices, and hospitals across four states.
By AccurKardia · Via Business Wire · April 15, 2026

AccurKardia, an innovator in ECG-based diagnostics technology, today announced results from a new study demonstrating that its AI-enabled ECG technology, AK-AVS™, can detect aortic stenosis years before patients require valve replacement and improve prediction of clinical outcomes. The study, “Validation and Longitudinal Trajectory Analysis of an AI-Based ECG Model for Aortic Stenosis: From Community Screening to Pre-TAVR Risk Stratification,” was led by principal investigator Dr. Matthew Segar, Electrophysiology Fellow at the Texas Heart Institute, and published in the European Heart Journal – Digital Health.
By AccurKardia · Via Business Wire · February 10, 2026

AccurKardia, an ECG-led diagnostics software company focused on transforming ECG data into a more powerful diagnostic tool and broad biomarker to improve patient outcomes and save lives, today announced U.S. Food and Drug Administration (FDA) 510(k) clearance and the commercial launch of its AccurECG™ Analysis System (v2.0) (K252361) (“AccurECG 2.0”). AccurECG 2.0 builds on AccurKardia’s previously FDA-cleared AccurECG platform, representing the next-generation evolution of the company’s enterprise-grade ECG interpretation system. With this milestone, the AccurECG platform has now secured two separate FDA 510(k) clearances.
By AccurKardia · Via Business Wire · January 8, 2026

AccurKardia, an innovator in ECG-based diagnostics technology, has announced the initiation of a multicenter clinical pilot of AK+ GuardTM, an FDA Breakthrough Device Designated AI-powered technology that enables the detection of hyperkalemia using Lead I ECG. The three-arm prospective pilot study will evaluate more than 150 patients across inpatient, outpatient and home settings in the U.S.
By AccurKardia · Via Business Wire · September 16, 2025

AccurKardia, an innovator in ECG-based diagnostics technology, today announced that its AccurECGTM software platform has been selected as winner of the “Best New Technology Solution for ECG” award in the 9th annual MedTech Breakthrough Awards. The program is conducted by MedTech Breakthrough, an independent market intelligence organization that recognizes the top companies, technologies, and products in the global digital health and medical technology market.
By AccurKardia · Via Business Wire · May 8, 2025
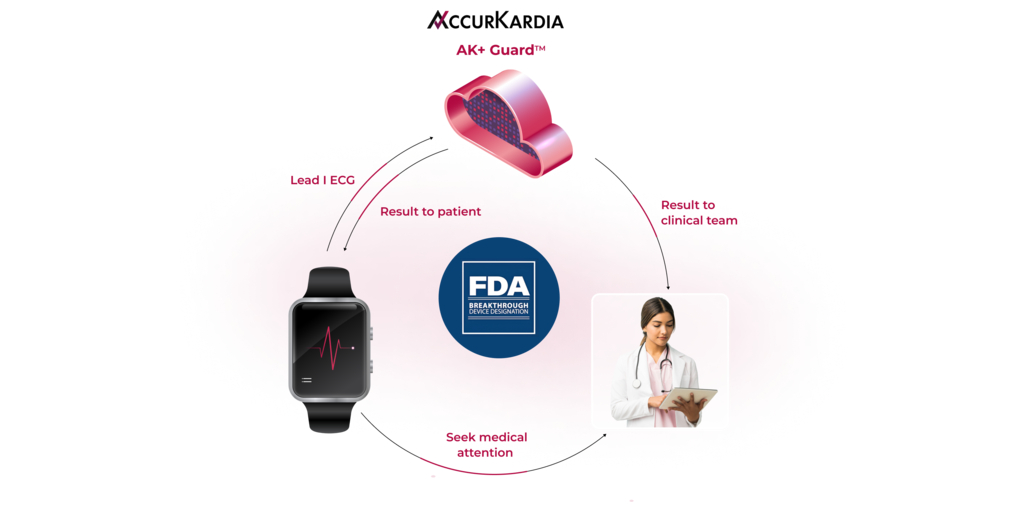
AccurKardia, an innovator in ECG-based diagnostics technology, has announced that it has received Breakthrough Device Designation by the U.S. Food and Drug Administration (FDA) for its ECG-based, AI-powered AK+ Guard™ hyperkalemia detection software. The investigational technology uses Lead I ECG data to alert patients and clinicians of moderate to severe episodes of hyperkalemia (excess potassium in the blood) that can lead to sudden cardiac arrest.
By AccurKardia · Via Business Wire · January 7, 2025

AccurKardia, an innovator in ECG-based diagnostics technology, has announced that its Aortic Valve Stenosis (AVS) ECG-based AI screening software has been granted Breakthrough Device Designation by the U.S. Food and Drug Administration (FDA). The company’s AVS screening software aims to leverage the ubiquity of the electrocardiogram (ECG) to identify potential cases of AVS within millions of ECGs already present in healthcare system electronic health records in order to help identify and prioritize which patients should receive echocardiograms for definitive diagnosis.
By AccurKardia · Via Business Wire · October 8, 2024

AccurKardia, an ECG-led diagnostics software company, today announced a multi-year agreement with Lucem Health, a leading provider of clinical AI solutions. The partnership paves the way for AccurKardia to integrate its FDA-Cleared ECG Interpretation Software Platform, AccurECG™ Analysis System (“AccurECG”), into Lucem Health Reveal, a family of solutions that identify patients who may be at higher risk of severe or chronic diseases.
By AccurKardia · Via Business Wire · November 9, 2023
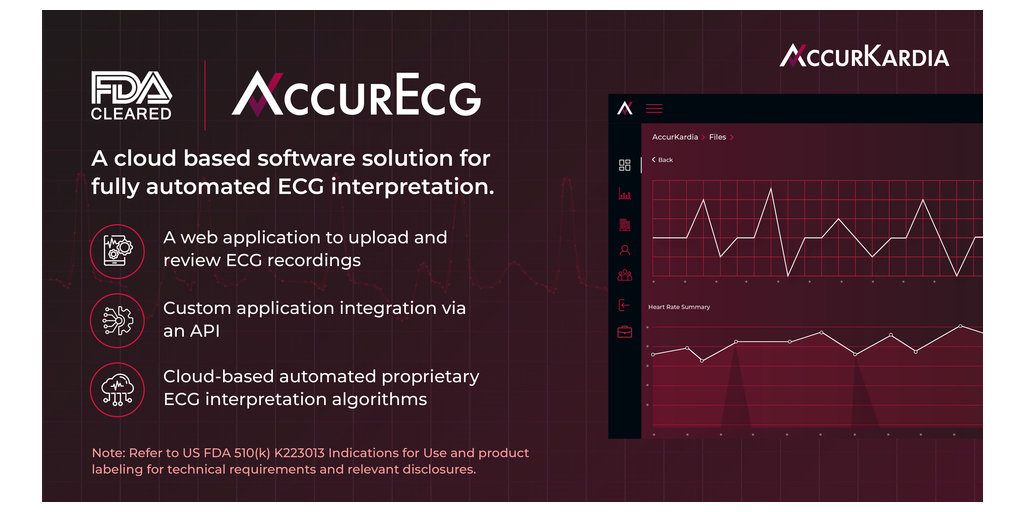
AccurKardia, a medical technology company delivering clinical-grade ECG interpretation software, announced today that its flagship product, the AccurECG™ Analysis System (“AccurECG” or the “System”), has been granted FDA 510(k) clearance. AccurECG™ is a cloud-based, device-agnostic and fully automated electrocardiogram (ECG) interpretation software platform.
By AccurKardia · Via Business Wire · July 11, 2023

AccurKardia, a software company that provides clinical-grade, device agnostic, automated electrocardiogram (ECG) analytics, announced today it will collaborate with Mawi, a medtech provider of medical grade wearables in the cardiac space, to integrate its proprietary ECG analytics into Mawi’s new cardiac monitoring watch. This collaboration highlights AccurKardia’s commitment to enabling world-class cardiac-care through remote patient monitoring solutions.
By AccurKardia · Via Business Wire · October 19, 2022

AccurKardia, a software company that provides clinical-grade, device agnostic, automated ECG (“electrocardiogram”) analytics, today announced the first study showcasing the clinical value of its AccurBeat platform. AccurBeat is AccurKardia’s platform for analysis of 30-seconds ECGs generated by wearable devices. The full study, conducted in collaboration by HITLAB (Healthcare Innovation & Technology Lab) and SUNY Downstate, can be found at this link from JMIR Publications.
By AccurKardia · Via Business Wire · October 11, 2022

AccurKardia, a software company that provides device-agnostic and automated ECG analytics, has joined the American Heart Association Center for Health Technology & Innovation’s (the Center) Innovators’ Network. The Center is focused on building and fostering health technology relationships to develop innovative and scalable solutions.
By AccurKardia · Via Business Wire · August 10, 2022
